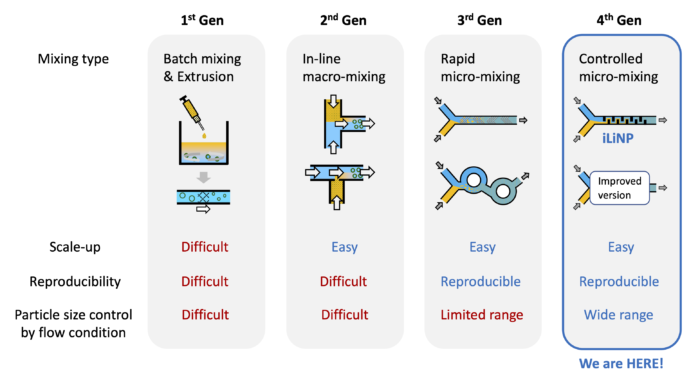
Microfluidic device “iLiNP” for lipid (polymer) nanoparticles manufacturing
The iLiNP (innovative Lipid Nanoparticles Production device) is the original microfluidic device suitable for manufacturing various kinds of lipid (or polymer) nanoparticles continuously from a lipid (or polymer) solution. The iLiNP consists of some inlets, outlet(s) and the channel having a lot of baffles.
Table of contents (Click to jump to each chapter)
- Mechanism of the nanoparticles formation in the iLiNP
- Advantage of the iLiNP
- Importance of particle size control
- Pending patents
1. Mechanism of the nanoparticles formation in the iLiNP
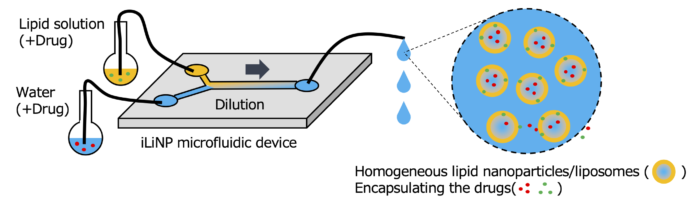
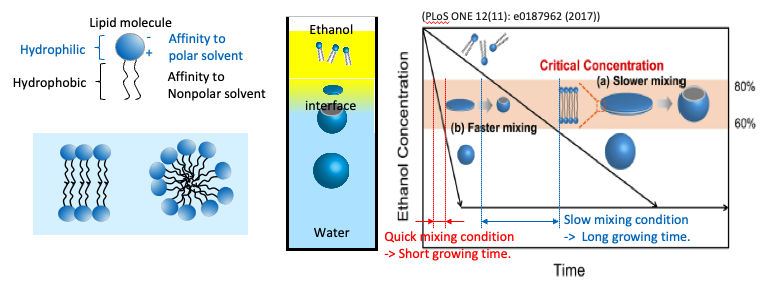
Lipid molecules (or amphiphilic polymers) are dissolved solely in an organic solvent such as alcohol. When the lipid solution meets water in the microfluidic device (iLiNP), the lipid solution is diluted at the liquid-liquid interface.
For example, in the case of POPC, a typical lipid molecule, when the Ethanol concentration falls below about 80%, the lipid molecules in the Ethanol begin to self-assemble with their hydrophilic groups facing outward. When the Ethanol concentration drops below about 60%, the self-assembled intermediate becomes spherical and stop growing.
If the mixing is gentle, the liquid-liquid interface is maintained for a long time, and the self-assembled intermediates near the interface grow larger until they reach their thermodynamically stable size.
If the mixing is vigorous, the intermediates cannot grow enough to form large-sized particles.
The shape of the flow channel in the iLiNP device is designed to change the dilution rate by flow conditions.
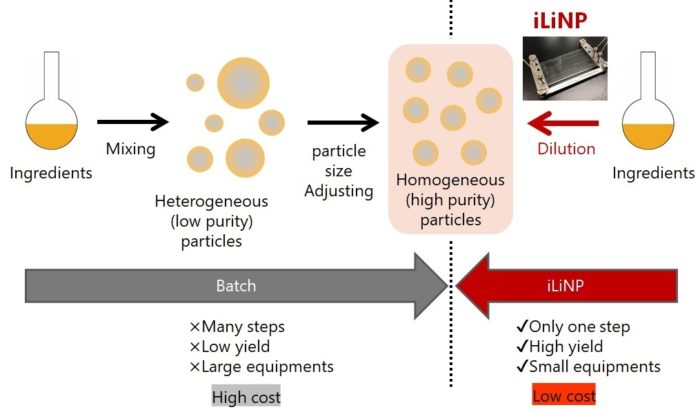
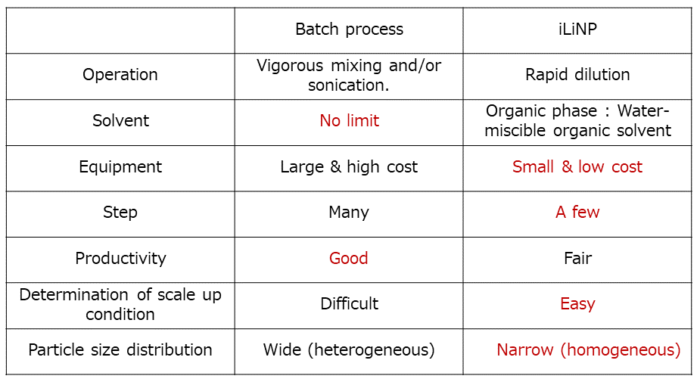
(1) Comparison between the batch process and the iLiNP
The iLiNP is a next-generation device having more advantages than the conventional batch process.
〇Reduction of manufacturing process.
〇High reproducibility of nanoparticle production and easy scale-up.
〇Small equipments.
〇As a result of the above, low-cost production of the nanoparticles.
(2) Comparison among the other microfluidic devices
The most valuable feature of the iLiNP is size-controlling ability of the nanoparticles.
(i) Preparation of large-size particles
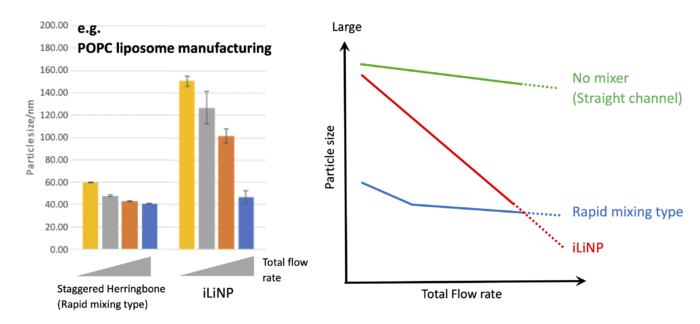
The reason why the iLiNP can easily produce large particles (>80 nm for a typical lipid composition), although the other company’s technologies cannot, is because of the different mixing strategy of the ingredients.
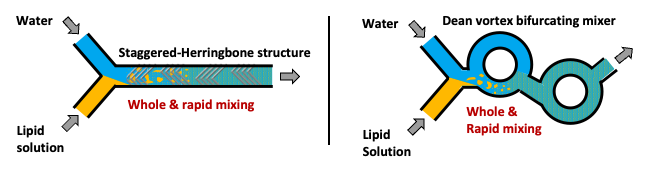
The other company’s technologies (shown in the figure above) adopt a strategy of whole and rapid mixing of the ingredients. The microchannel structures and the flow conditions are designed to achieve the mixing.
As a result, stable and large amount production of the nanoparticles is possible, but larger particle is not generated due to rapid dilution caused by the intense mixing.
On the other hand, the iLiNP (shown in the above figure) adopt a strategy of no-whole & no-rapid mixing of the ingredient at a typical flow condition.
As a result, the liquid-liquid interface, where self-assembly of the lipids proceeds, is maintained for a long time, and large particles can be obtained (<200 nm for a typical lipid composition, <500 nm if the lipid composition is also modified).
(ii) Preparation of small-size particles
Another feature of the iLiNP is a key point for preparation of small particles using the iLiNP.
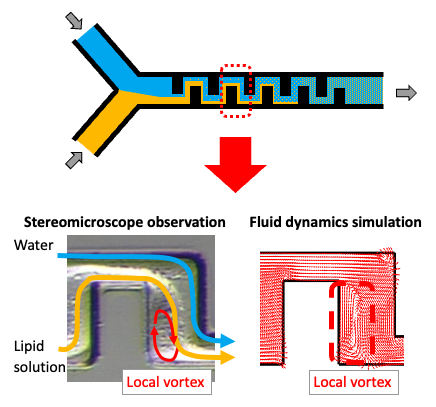
Under a high flow condition, the channel structure adopted in the iLiNP generates a local vortex (local mixing) in the flow channel while maintaining the interface as shown in the above figure.
As a result, local dilution is accelerated only under the high flow rate conditions, and small particles can be obtained.
The efficiency is so high that even smaller particles than those produced by other technologies can be obtained at a flow rate of about 1/10 that of other technologies.
Change in the flow in response to the flow rate. (Water/Ethanol system. Ethanol phase is stained by red dye.)

The above graph summarizes the features of the microfluidic devices.
Other company’s’ conventional mixer devices focus on quick mixing, and as a result, they can produce small particles even at a low flow rate condition. But the range of particle size change in response to flow rate change is narrow.
On the other hand, the iLiNP does not induce rapid mixing at a low flow rate, and the change in particle size as a function of flow rate is gradual. As the flow rate is increased, a local vortex is appeared and dilution is accelerated, even in the flow rate range where mixing and dilution performance is normally saturated. As a result, the range of particle size change in response to flow rate change is very wide.
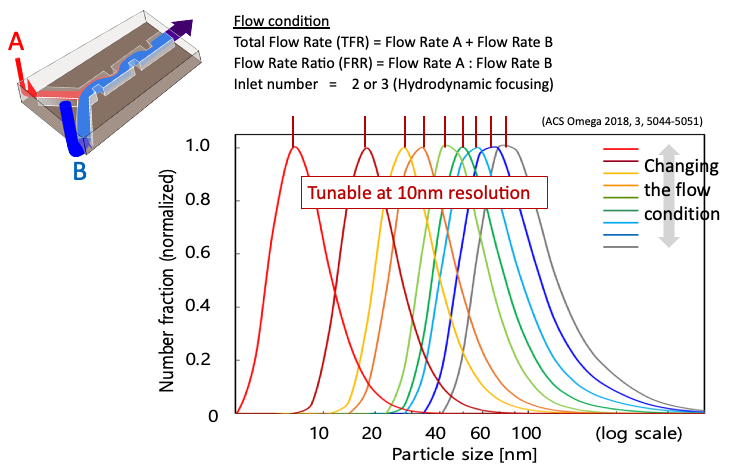
The above graph is an example of the particle size control. The iLiNP was used to control the size of nanoparticles using POPC, a typical lipid. This indicates that the iLiNP enables to tune the size at 10nm resolution by flow conditions.

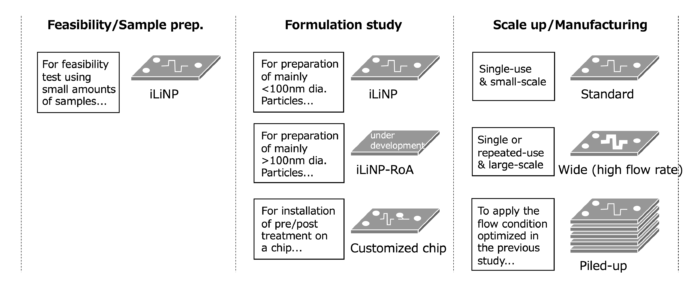
It is easy to scale up the manufacturing process with the iLiNP. Various materials can be used for the flow device due to the simple flow channel structure. For example, for scaling up by increasing the flow rate, the device made of stainless steel (upper left photo) or glass devices can be used because of their resistance to pressure. If you want to receive the benefit of the iLiNP’s “particle size control by flow rate control” feature, you can scale up by increasing the number of devices (piling up and numbering up) where the optimum flow rate per device is maintained (upper middle photo, upper right figure). Generally, there is no need to reinvestigate the manufacturing conditions. In addition, since the number of manufacturing facilities (devices) can be easily increased or decreased according to the required volume, the risk of excessive investment in manufacturing facilities can be reduced.
3. Importance of particle size control
Recent research papers have shown that the biological activity of nanoparticle formulations can vary by as little as 10 nanometers in size.
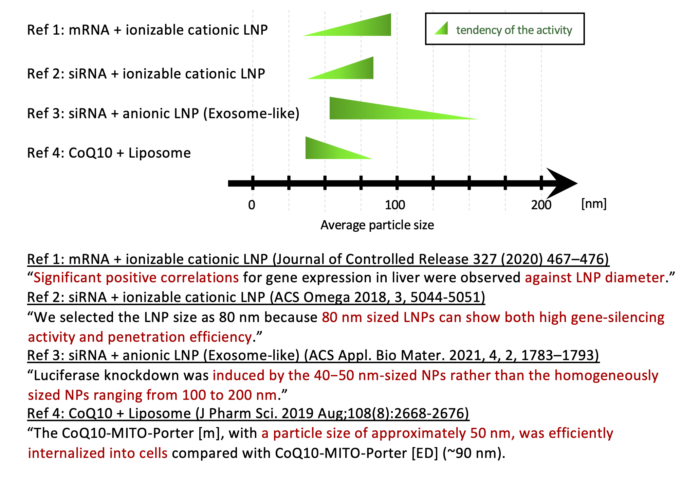

The above is just some examples, and we expect to accumulate more similar knowledge as our technology spreads.
With our iLiNP device, you can get to the “maximization of efficacy through precise control of particle size” before anyone else.
- Flow channel structure and lipid particle or micelle formation method using same.
Patent No. : JP6942376 (Japan)
Application No. : PCT/JP2018/015550 -> transfer into US, EP, CN, CA
107112803 (Taiwan)
Applicant : NATIONAL UNIVERSITY CORPORATION HOKKAIDO UNIVERSITY
Filing Date : Apr 13, 2018 (Priority date : Apr 13, 2017)
License : Exclusive license granted to Lilac pharma Inc. - Production method of particle containing solution
Application No. : JP2020-556107 (Japan)
Applicant : NATIONAL UNIVERSITY CORPORATION HOKKAIDO UNIVERSITY
Filing Date : Nov 6, 2019 (Priority date : Nov 9, 2018)
License : Exclusive license granted to Lilac pharma Inc. - Flow channel structure and self-assembled molecular particles formation method using same.
Application No. : JP2022-017619 (Japan)
Applicant : NATIONAL UNIVERSITY CORPORATION HOKKAIDO UNIVERSITY
Filing Date : Feb 8, 2022 (Priority date : Feb 8, 2022)
License : Exclusive license granted to Lilac pharma Inc. - Manufacturing method of emulsified fragrance
Application No. : JP2020-159344 (Japan)
Applicant : Lilac pharma Inc.
Filing Date : Sep 24, 2020 (Priority date : Sep 24, 2020)

