1. On-site manufacturing as a new nanoparticles supply concept
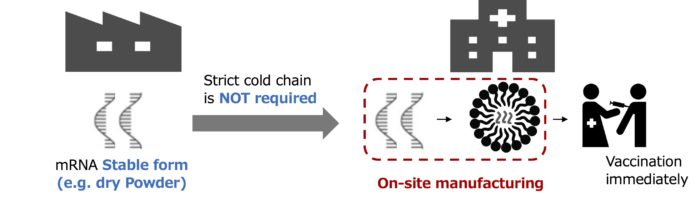
Many nanoparticle formulations are unstable and require refrigeration or freeze-drying for long-distance transportation or storage, which can be costly and difficult in developing countries.
We have come up with a new idea for supplying nanoparticle drugs and vaccines called “on-site manufacturing”. In the concept of the on-site manufacturing, we can keep the ingredients such as lipids and RNAs at pharmacies and use microfluidic devices to quickly and easily manufacture the nanoparticle drugs and vaccines right before they are administrated to patients. Use of the micro fluidic device ensures high-quality and reproducible results.
In the case of lipid nanoparticles, the ethanol used in the on-site manufacturing is considered low toxicity (Class 3) according to pharmaceutical guidelines on residual solvents. This means that safe formulations can be provided without the need to remove ethanol completely, by carefully considering administration methods and dosages that comply with safety guidelines. In fact, mouse experiments have shown that vaccine efficacy can be maintained even with a nanoparticle vaccine that contains ethanol (JP 2023-025886).
To implement the on-site manufacturing, we have developed a prototype small-volume manufacturing device that uses sterile, single-use products for the entire liquid delivery line. We have also started user hearings to gather feedback and improve the device.

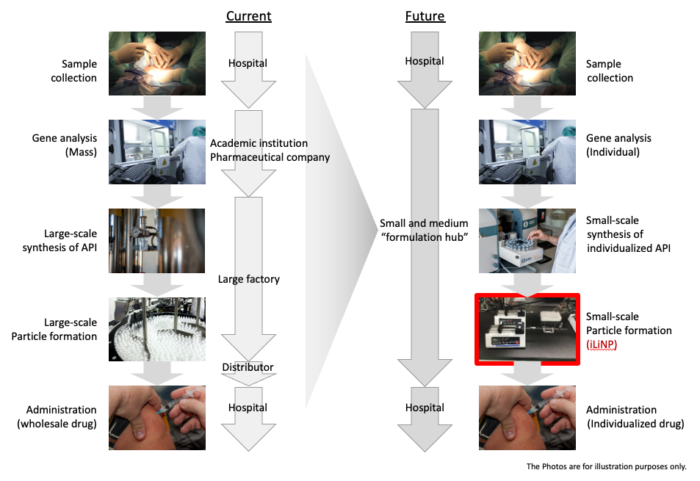
2. On-site formulation can be used to produce small-volume, high-variety “tailor-made” drugs using microfluidic devices.
The microfluidic devices are especially suitable for personalized nanoparticle formulations for individual patients in personalized medicine. As a result, it is expected that mass production facilities in factories will no longer be necessary, and on-site manufacturing in pharmacies will become the preferred method. We are actively researching and developing new technologies to stay ahead of the next generation of drug formulation trends.